
The electronic configuration of Potassium will be 1s2 2s2 2p6 3s2 3p6 4s1. How do you write the electron configuration for Potassium? In the periodic table, elements with chemical properties that are similar are grouped together in the same group. The number of valence electrons an atom has determines its location in the period: the element with the electron configuration of ' Ne'3's'2 3'p'4 has six valence electrons, so within. Groups are the vertical columns of the periodic table that are counted from left to right, from 1 to 18. Valence electrons relate to the position of elements within the groups and periods of the periodic table, and also their position within blocks. What is the electronic configuration of Potassium 19? Determine the element’s group number and period number by referring to the periodic table. What is the boiling Point of Potassium in Kelvin?īoiling Point of Potassium in Kelvin is 1032 K. Melting Point of Potassium in Kelvin is 336.53 K. Group 1 elements have just one valence electron while group 18 elements have eight (except for helium which has only two electrons total). What is the melting Point of Potassium in Kelvin? 1: Periodic table by Dmitri Mendeleev, 1871. After the development of quantum mechanics, it was shown that the halogens all had seven valence electrons, supporting their original placement into the same group on Mendeleev's periodic table. What is the boiling Point of Potassium?īoiling Point of Potassium is 1032 K. As a gas or vapor, the halogens all had a pungent odor. Potassium has 19 electrons out of which 1 valence electrons are present in the 4s1 outer orbitals of atom. 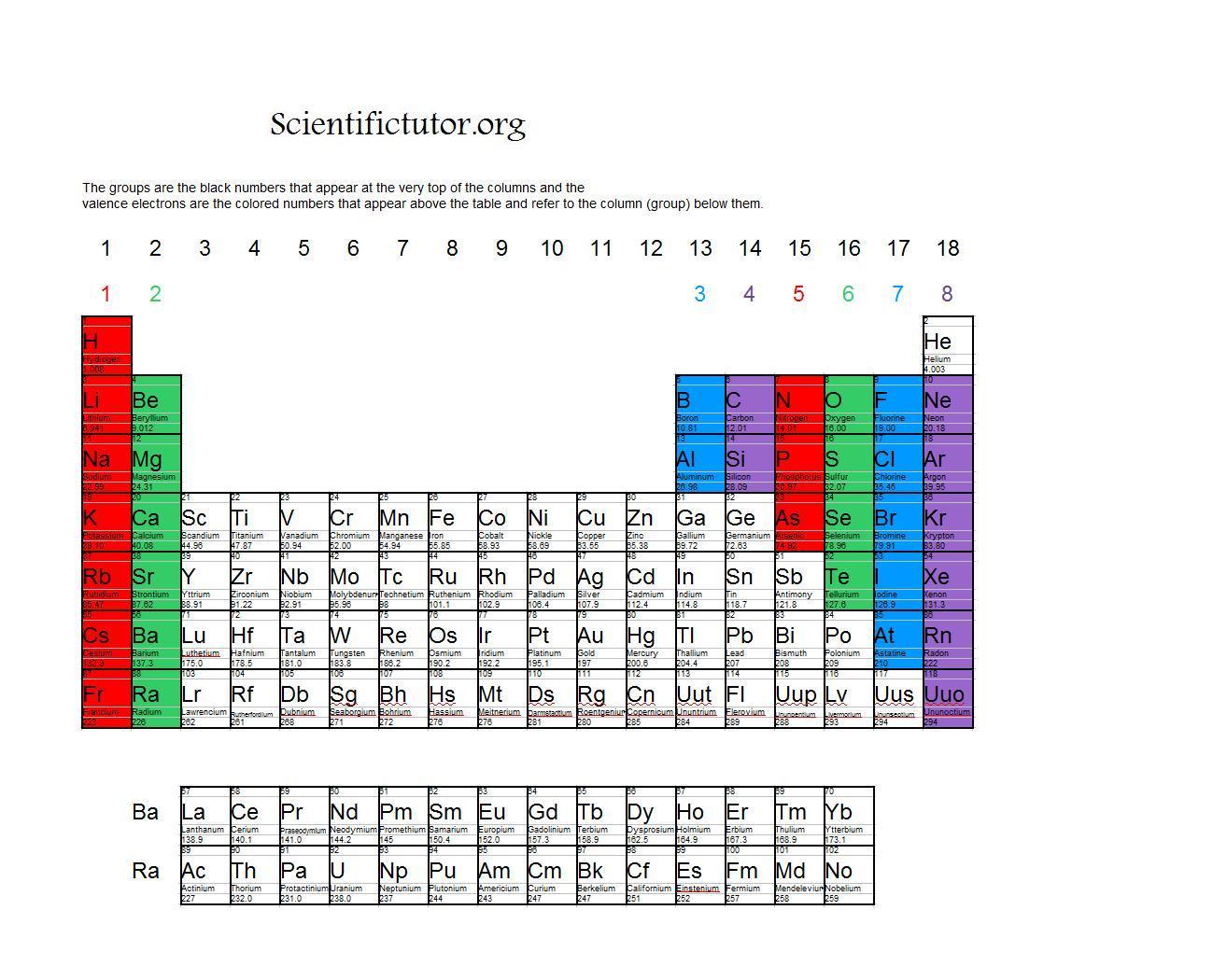
How many valence electrons does a Potassium atom have? The element Potassium was discovered by H. You can read the number of valence electrons that an element has directly from the periodic table, using the old-style group numbers (Roman numeral plus. It is located in group 1 and period 4 in the modern periodic table. Potassium is the 19 element on the periodic table. Potassium is a chemical element with the symbol K and atomic number 19. What is the position of Potassium in the Periodic Table? Potassium is a chemical element with symbol K and atomic number 19. To form abbreviated notation of electronic configuration, the completely filled subshells are replaced by the noble gas of the preceding period in square brackets. 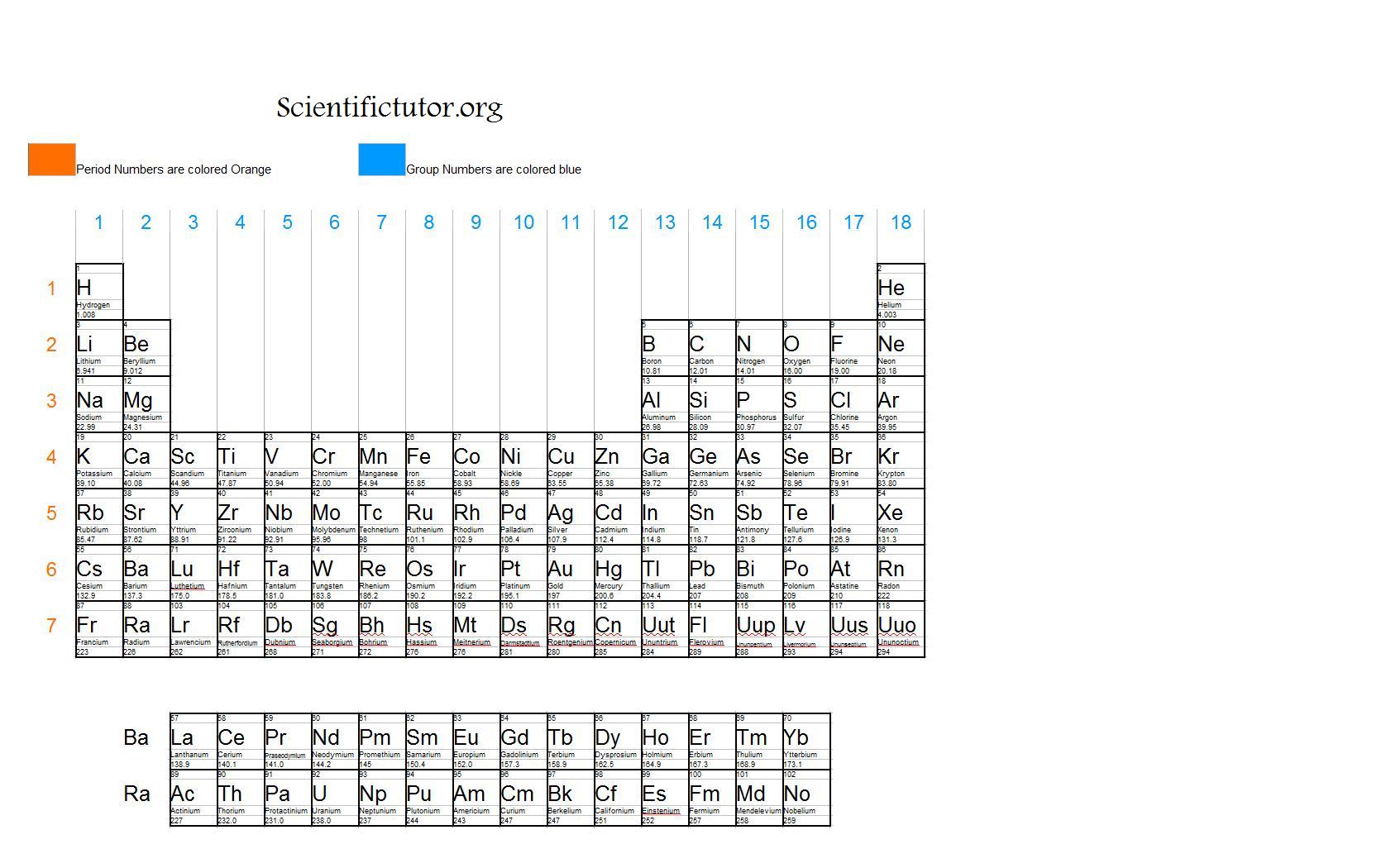
The abbreviated electronic configuration of Potassium is 4s1. What is the abbreviated electronic configuration of Potassium? The electronic configuration of Potassium is 1s2 2s2 2p6 3s2 3p6 4s1. Questions Tips & Thanks Want to join the conversation Sort by: Top Voted Manahil Ahmed 9 years ago If helium is in group 8 why does it not have 8 valence electrons. What is the electronic configuration of Potassium? How to determine the number of valence electrons and draw Lewis structures for main group elements starting from the electron configuration. Potassium Thermal Properties - Enthalpies and thermodynamics Optical Properties of Potassium Refractive IndexĪcoustic Properties of Potassium Speed of Sound Potassium Magnetic Properties Magnetic Type Potassium Heat and Conduction Properties Thermal Conductivity Refer to table below for the Electrical properties ofPotassium Electrical Conductivity Hardness of Potassium - Tests to Measure of Hardness of Element Mohs Hardness \) provides an alternative method for determining the electron configuration.Refer to below table for Potassium Physical Properties DensityĠ.856 g/cm3(when liquid at m.p density is $0.828 g/cm3)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
